On a prior thread, Gail shared a story about goats and an accidental improvement in her health:
https://chiefio.wordpress.com/2021/08/24/a-useful-page-on-chinese-wuhan-covid/#comment-149398
Gail Combs says:
24 August 2021 at 11:09 pm (Edit)
Hi guys,
[…]
Well three months ago it was time to worm and inoculate my goats, all 25. Since I had breathing difficulties not to mention the fun of trying to rope goats with a right handed lariat with my left hand, I decided to go for my three big boys first. (100+ pounds each) I got to hold their heads up and still while Hubby medicated. They were NOT happy about getting Moxidectin sheep drench pumped down their throats and ended up spitting the wormer and goat drool all over my hands. Four or five hours later I got to remove the drool from 25 goats….I HAVE NOT HAD ‘ASTHMA’ SINCE!!! And my O2 is now 96%-97% while my pulse has gone from the low hundreds to in the sixties.
Moxidectin and ivermectin inhibit SARS-CoV-2 replication in Vero E6 cells but not in human primary airway epithelium cells but in my case it sure looks like it took care of what ever was happening in my lungs.
[…]
Then AC Osborn pointed out a movement by the most bought off to ban Ivermectin from use for Chinese Wuhan Covid:
AC Osborn says:
2 September 2021 at 5:25 pm (Edit)
The AMA calls for Ivermectin to be banned for treating COVID.
H/T to both!
Also note that in California, buying ivermectin for cattle is now on a “restricted list” of some kind and you get to give you ID, phone# etc to them… So I just bought a lifetime supply of 2.5 liters… (or close to a lifetime supply I think)
But, on the off chance it starts to be harder to find / buy, I though it might be worth a re-visit of the story of the goats and a comparison of the two molecules. (Hey, if they want to start a game of “Whack A Mole” I’m game…)
The link in Gail’s comment points to a PDF about the two drugs.
Moxidectin and ivermectin inhibit SARS-CoV-2 replication in Vero E6 cells but not in human primary airway epithelium cells
Nilima Dinesh Kumar1,2#, Bram Ter Ellen2#, Ellen M. Bouma2#, Berit Troost2, Denise P. I. van de Pol2, Heidi H. van der Ende-Metselaar2, Djoke van Gosliga4, Leonie Apperloo3 Orestes A. Carpaij5, Maarten van den Berge5, Martijn C. Nawijn3, Ymkje Stienstra6, Izabela A RodenhuisZybert 2#, Jolanda M. Smit2#*
1. Department of Biomedical Sciences of Cells & Systems, University of Groningen, University Medical Center Groningen, The Netherlands
2. Department of Medical Microbiology and Infection Prevention, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands
3. Department of Pathology and Medical Biology, University Medical Center Groningen, University of Groningen, GRIAC Research Institute, 9700 RB Groningen, The Netherlands
4. Department of Pediatrics, Beatrix Children’s Hospital, University Medical Center Groningen, University of Groningen, GRIAC Research Institute, 9700 RB Groningen, The Netherlands.
5. Department of Pulmonary Diseases, University Medical Center Groningen, University of Groningen, GRIAC Research Institute, 9700 RB Groningen, The Netherlands
6. Department of Internal Medicine/Infectious Diseases, University of Groningen, University Medical Center Groningen, The Netherlands
Not lightweights. But Universities can have agenda…
I’m going to bold a few lines.
Abstract
Antiviral therapies are urgently needed to treat and limit the development of severe COVID-19 disease. Ivermectin, a broad-spectrum anti-parasitic agent, has been shown to have anti-SARSCoV-2 activity in Vero cells at a concentration of 5 µM. These in vitro results triggered the investigation of ivermectin as a treatment option to alleviate COVID-19 disease. In April 2021, the World Health Organization stated, however, the following: “the current evidence on the use of ivermectin to treat COVID-19 patients is inconclusive”. It is speculated that the in vivo concentration of ivermectin is too low to exert a strong antiviral effect. Here, we performed a head-to head comparison of the antiviral activity of ivermectin and a structurally related, but metabolically more stable, moxidectin in multiple in vitro models of SARS-CoV-2 infection, including physiologically relevant human respiratory epithelial cells. Both moxidectin and ivermectin exhibited antiviral activity in Vero E6 cells. Subsequent experiments revealed that the compounds predominantly act on a step after virus cell entry. Surprisingly, however, in human airway-derived cell models, moxidectin and ivermectin failed to inhibit SARS-CoV-2 infection, even at a concentration of 10 µM. These disappointing results calls for a word of caution in the interpretation of anti-SARS-CoV-2 activity of drugs solely based on Vero cells. Altogether, these findings suggest that, even by using a high-dose regimen of ivermectin or switching to another drug in the same class are unlikely to be useful for treatment against SARSCoV-2 in humans.
A lot of weaselly words in there… speculated, suggest. Since Chinese Wuhan Covid is known to attack all sorts of cells all over the body, having one test culture that’s less affected by a treatment seems weak tea.
I find this bit more useful:
[…] ivermectin was found to exert significant antiviral activity towards SARS-CoV-2 in Vero E6 cells (Fig.1). In presence of 10 µM ivermectin, infectious virus particle production was reduced to 1.9 ± 0.8 x10^2 PFU/mL, which corresponds to a reduction of more than 99.9% when compared to the EtOH control (Fig. 1A). In presence of 10 µM of moxidectin, the infectious virus titer was reduced to 4.3 ± 2.0 x10^2 PFU/mL which also corresponds to a reduction of 99.9% when compared to the EtOH control (Fig. 1A).
So the two tend to act the same (whatever that action might be…) Then, further down after testing various parts of the virus replication cycle:
Collectively, these results indicate that both moxidectin and ivermectin directly interfere with the viral infectious replication cycle in cells. This shows that the compounds either act at the early stages of RNA replication/translation (within 2-4 h post-infection) or that the compounds interfere with late stages of virus assembly/secretion (i.e. needs to be present for more than 4 h to exert its antiviral effect)
So it acts early and is likely best as a prophylactic or very quick and early treatment. Plus both work to inhibit virus replication.
Also, once again, we have a rubber ruler in use. The Vaccines are considered just dandy and being promoted for prevention of body sickness while happily letting the virus infect and reproduce and spread in the vaccinated via lungs / throat / mucosa; while these two drugs are being dissed as useless for not stopping replication in lung cells.
3.3 Moxidectin and ivermectin do not exhibit anti- SARS-CoV-2 activity in Calu-3 and primary human bronchial epithelial cells
We next sought to validate the antiviral potential of moxidectin and ivermectin in the human lung epithelial Calu-3 cells, cells which have previously been shown to support SARS-CoV-2 replication (Chu et al., 2020). Prior to infectivity assays, the cellular cytotoxicity of both compounds at 5 and 10 µM was determined using the MTS assay. At these concentrations, no cytotoxicity was observed (Fig. S3). Accordingly, we proceeded by infecting Calu-3 cells with SARS-CoV-2 at MOI 1 in the presence or absence of 5 µM and 10 µM of moxidectin or ivermectin. In the absence of the drugs (NT condition), infection led to a release of on average, 1.5 ± 2.3 x10^4 PFU/mL at 8 hpi (Fig. 3A). A comparable titer was observed for the EtOH control (1.7 ± 2.5 x10^4 PFU/mL). To our surprise, however, no significant reduction in viral titers was observed following infection in presence of moxidectin and ivermectin, indicating that at these experimental conditions, the compounds did not exhibit an antiviral effect in Calu-3 cells.
Go figure…
OK, for my purposes, I’m mostly interested in the statements showing these two drugs are very similar with very similar actions and results; and that they are antiviral at least in some cells.
So what are these things?
In my reply to Gail in the earlier discussion I cited some text from :
https://www.msdvetmanual.com/pharmacology/anthelmintics/macrocyclic-lactones
[…]
@Gail:That is a marvelous find and adds One More Tool to the ‘ol tool box. I just looked up Moxidectin in the wiki and it looks to me like an Ivermectin with the long tail chopped off and the “do-dads” in the upper right a bit different. Same class of compounds, BTW. I suspect there’s a whole bucket full of related stuff that will all plug up the end of the Spike Protein to greater or lesser degrees. Macrocyclic Lactones. I’m bolding a couple of bits.
Macrocyclic Lactones
By Jozef Vercruysse , DVM, Ghent University;
Edwin Claerebout , DVM, PhD, DEVPC, Department of Virology, Parasitology and Immunology, Faculty of Veterinary Medicine, Ghent UniversityLast full review/revision Sep 2014 | Content last modified Oct 2014
The macrocyclic lactones (avermectins and milbemycins) are products or chemical derivatives of soil microorganisms belonging to the genus Streptomyces. The avermectins in commercial use are ivermectin, abamectin, doramectin, eprinomectin, and selamectin. Commercially available milbemycins are milbemycin oxime and moxidectin. The macrocyclic lactones have a potent, broad antiparasitic spectrum at low dose levels. They are active against many immature nematodes (including hypobiotic larvae) and arthropods. The published literature contains reports of use to treat infections of >300 species of endo- and ectoparasites in a wide range of hosts. Moreover, a single therapeutic dose can persist in concentrations sufficient to be effective against new nematode infections for prolonged periods after treatment.
The macrocyclic lactones are well absorbed when administered PO or parenterally; the pour-on formulations exhibit greater variability. Regardless of the route of administration, macrocyclic lactones are extensively distributed throughout the body and concentrate particularly in adipose tissue. However, the route of administration and formulation may affect the drug pharmacokinetics. The residence time of macrocyclic lactones administered SC may also be influenced by the body condition of the animal.
Effective levels are reached in the GI system, lungs, and skin regardless of the route of administration. There is, however, a very complex interaction between pharmacokinetic compartments and the quantitative and qualitative availability of drug/metabolite in one compartment. For example, the association of macrocyclic lactones with digesta affects absorption; systemic availability and elimination of ivermectin given PO may differ significantly with feed quantity or composition in sheep. Also, the practice of feed withdrawal before PO treatment broadens the pharmacokinetic profile, significantly increasing anthelmintic efficacy.So if the Idiots In Charge ban one, we’ve now got a shopping list of others to explore…
Here’s some bits from the wiki on them:
https://en.wikipedia.org/wiki/Ivermectin
Ivermectin is a medication that is used to treat parasite infestations. In humans, this includes head lice, scabies, river blindness (onchocerciasis), strongyloidiasis, trichuriasis, ascariasis, and lymphatic filariasis.
[…]
Ivermectin was discovered in 1975 and came into medical use in 1981.[14][15] It is on the World Health Organization’s List of Essential Medicines.[16] Ivermectin is FDA-approved as an antiparasitic agent. In 2018, it was the 420th most commonly prescribed medication in the United States, with more than one hundred thousand prescriptions.
[…]Adverse effects
Ivermectin is considered relatively free of toxicity in standard doses, that is, around 300 ug/kg. Based on the data drug safety sheet for ivermectin (New Drug Application Identifier: 50-742/S-022), side effects are uncommon.
[…]
One concern is neurotoxicity after large overdoses, which in most mammalian species may manifest as central nervous system depression, ataxia, coma, and even death, as might be expected from potentiation of inhibitory chloride channels.Since drugs that inhibit the enzyme CYP3A4 often also inhibit P-glycoprotein transport, the risk of increased absorption past the blood-brain barrier exists when ivermectin is administered along with other CYP3A4 inhibitors. These drugs include statins, HIV protease inhibitors, many calcium channel blockers, lidocaine, the benzodiazepines, and glucocorticoids such as dexamethasone.
During the course of a typical treatment, ivermectin can cause minor aminotransferase elevations and, rarely, mild clinically apparent liver disease.
[…]
ChemistryAvermectins produced by fermentation are the chemical starting point for Ivermectin
Fermentation of Streptomyces avermitilis yields eight closely related avermectin homologues, of which B1a and B1b form the bulk of the products isolated. In a separate chemical step, the mixture is hydrogenated to give ivermectin, which is an approximately 80:20 mixture of the two 22,23-dihydroavermectin compounds.History
The avermectin family of compounds was discovered by Satoshi Ōmura of Kitasato University and William Campbell of Merck. In 1970, Ōmura isolated unusual Streptomyces bacteria from the soil near a golf course along the south east coast of Honshu, Japan. Ōmura sent the bacteria to William Campbell, who showed that the bacterial culture could cure mice infected with the roundworm Heligmosomoides polygyrus. Campbell isolated the active compounds from the bacterial culture, naming them “avermectins” and the bacterium Streptomyces avermitilis for the compounds’ ability to clear mice of worms (in Latin: a ‘without’, vermis ‘worms’). Of the various avermectins, Campbell’s group found the compound “avermectin B1” to be the most potent when taken orally. They synthesized modified forms of avermectin B1 to improve its pharmaceutical properties, eventually choosing a mixture of at least 80% 22,23-dihydroavermectin B1a and up to 20% 22,23-dihydroavermectin B1b, a combination they called “ivermectin”.
Ivermectin was introduced in 1981. Half of the 2015 Nobel Prize in Physiology or Medicine was awarded jointly to Campbell and Ōmura for discovering avermectin, “the derivatives of which have radically lowered the incidence of river blindness and lymphatic filariasis, as well as showing efficacy against an expanding number of other parasitic diseases”.
Note that large doses are used for the various parasites it is used to cure and the cautions are mostly about side effects of a large die-off of parasites inside you or from really large overdoses.
Here’s a comparison of the Ivermectin molecule with the Moxidectin molecule:
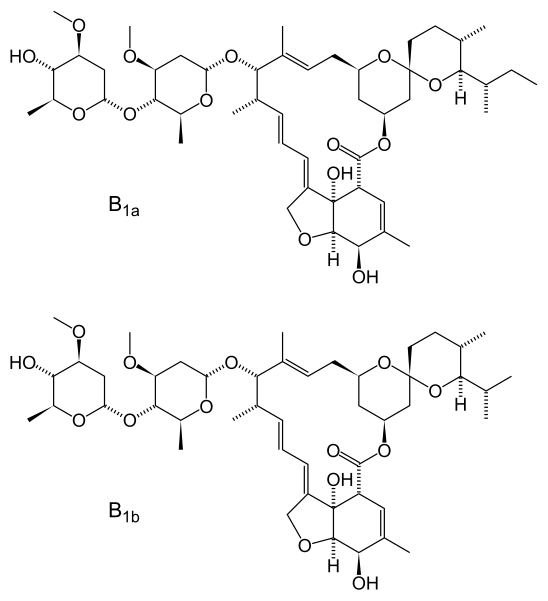
Ivermectin Molecules

Moxidectin Molecule
To my eye, the two drugs look to share a common center part with a couple of minor diddles on the bottom and right, and Ivermectin adds a big tail to the left. I’d speculate that the common chunk is the major active part in both.
https://en.wikipedia.org/wiki/Moxidectin
Medical uses
Moxidectin was approved for onchocerciasis (river-blindness) in 2018 for people over the age of 11 in the United States based on two studies. There is a need for additional trials, with long-term follow-up, to assess whether moxidectin is safe and effective for treatment of nematode infection in children and women of childbearing potential. Moxidectin is predicted to be a helpful to achieve elimination goals of this disease.Dogs: Prevention of heartworm. In combination with imidacloprid to treat sarcoptic mange.
Horses: Treatment of parasites including Strongylus vulgaris, and stomach bots such as Gasterophilus intestinalis.
Cattle: Treatment of parasites such as the gastrointestinal nematode Ostertagia ostertagi, and the lungworm Dictyocaulus viviparus.
Sheep: Treatment of the nematodes Teladorsagia circumcincta and Haemonchus contortus.
Nematodes can develop cross-resistance between moxidectin and other similar parasiticides, such as ivermectin, doramectin and abamectin.Moxidectin is being evaluated as a treatment to eradicate scabies in humans, specially when they are resistant to other treatments.
So there’s a couple of more to investigate should Whack-A-Mole be on the cards. I note in passing that Moxidextin is being used in humans.
Chemistry
Moxidectin, a macrocyclic lactone of the milbemycin class, is a semisynthetic derivative of nemadectin, which is a fermentation product of the bacterium Streptomyces cyanogriseus subsp. noncyanogenus.
History
In the late 1980s, an American Cyanamid Company agronomist discovered the Streptomyces bacteria from which moxidectin is derived in a soil sample from Australia. Two companies filed patents for moxidectin: Glaxo Group and the American Cyanamid Company; in 1988, all patents were transferred to American Cyanamid. In 1990, the first moxidectin product was sold in Argentina.
For human use, moxidectin was approved by the United States Food and Drug Administration in June, 2018 for the treatment of onchocerciasis in adults and adolescents aged 12 and older. This is the first human approval worldwide. The license holder is the nonprofit biopharmaceutical company Medicines Development for Global Health.
So if Ivermectin becomes unobtainium, there’s some alternatives to look for…
Note that none of this is medical advice. I am not a Doctor nor do I play one on the internet. This is for educational and discussion purposes only. You do you, I’ll do me.

I knew I had been convinced of Ivermectin for a long time and bought some veterinary products when I first became interested. Out of curiosity I checked and I first bought veterinary Ivermectin in early April 2020. What took the rest of you so long? Also, the price of veterinary Ivermectin has more than doubled since then, IF you can get it. You do have to pay attention if you’re getting veterinary products. Some have additional medications (could be very bad). Some have other non-medicinal things added (check carefully). Some are formulated for different modes of use (DO NOT DRINK IVERMECTIN FORMULATED FOR TOPICAL USE – it could kill you, but using it topically should be OK). All of them you have to figure out what the equivalent volume is for the appropriate human dosage for your weight.
“It’s open war on Ivermectin now”
https://joannenova.com.au/2021/09/its-open-war-on-ivermectin-now/#comment-2462943
And O/T BUT
“What do you do when China steals your entire company out from under you?”
https://bayourenaissanceman.blogspot.com/2021/09/what-do-you-do-when-china-steals-your.html
ARM chips
@AnnieM:
Well, I had a 1/2 full 250 ml bottle of it as “Cattle Drench” pour on from the start… left over from a Rabbit Cure for wry neck / e. cuniculi…
So I’ve had it since 2011 ;-)
When it was used up, I bought another 250 ml bottle at the Feed Store… then when that was used up, a couple of months back, bought a 2.5 L jug. At my present usage rate for the family, I think it works out to a 5 year supply…
Did take me a couple of months to get around to using some of it though… I used it as a “post symptom immediate treatment” for about 6 to 8 months (applied only when I had snivels or some scratchy throat) then swapped to once a week prophylaxis in the mode of the FLCCC when I started working part time for a Dentist a few months back… where by definition there’s a lot of people without a mask over their mouth…
Overall, I think I’ve been using it about 1.5 years now, more or less. I suppose I could dig through the postings and find my first mention of it…
BTW, I’ve now gone coast to coast twice, spent many days maskless in Walmarts, Gas Stations, bars & restaurants from California to Florida and up the East Coast and in Chicago (and all points in between…) including a couple of Bars in Florida with a big crowd of folks and no mask in sight. All with “no bad thing”… so I think I’m an existence proof that it works. Especially given that I’m in the “old and grey” group that is supposedly a prime target.
But whatever. Maybe I’m just Superman with a Super Immunity ;-)
Looking at various Sheep Oral products, I found they vary a lot in the “other stuff” in the jug. Many contain selenium so you would need to compute the dose to assure there was not a selenium toxicity issue (it doesn’t take much… one Brazil Nut a day is how much you need…). Others have a LOT of some solvents in them that might require research. This MSDS looks like a pretty clean one:
Link:http://www.evs.co.nz/assets/Material-Safety-Data-Sheets/Zoetis/Cydectin-oral-drench-for-sheep.pdf
So only Benzyl Alcohol to worry about. It has an LD-50 of 1.2 g/kg in rats per the wiki:
https://en.wikipedia.org/wiki/Benzyl_alcohol
If we are at all like rats, that’s up about 120 grams for a 100 kg me. As the percent is 3.8% and the dose is about 1ml / 5 kg of sheep, that’s 20 ml for 100 kg that’s about 0.76 gram total in the dose, or about 0.006 of the LD-50. It is used in various skin products and drugs, so clearly not a big toxin.
So looks like it’s biggest issue is just that you might have an allergy to it, so test a small bit first. Then keep it out of your eyes and away from babies.
Looks like a reasonable “Plan B” (or C or D…) should Ivermectin become unobtanium…
@Another Ian:
We have a W.O.O.D. – Weekly Occasional Open Discussion – page for things that are Off Topic in topic oriented pages. Please use it.
Just out of curiosity, I took a peek at one of my dogs meds.
I live in a very rural area but saw a five liter bottle of Ivermectin cattle drench at the Co-Op today. Price? $79.
@Ossqss that’s a very small amount of Ivermectin. To put it in perspective you’d have to take somewhere on the order of 100 of those chews to get a reasonable dose. Then you’d have to worry about what Pyrantel is and what it would do to you.
E.M.
The current Wood is around the corner. Knowing your interest in ARM chips was why I put that there
Be sure and check the ingredients in your Ivermectin for animals. My bro bought some that had a “not so good for humans ingredient” in it according to his pharmacist friend.
@Another Ian:
You can always scroll down to “categories” and click on “W.O.O.D”. The top one is the newest…
Not a big deal, but it is nice to do so I can have some hope of finding some item later. (Why would I ever look for ARM chips under a posting clearly about drugs?…)
@John S Howard, Jr:
I’ve found that the online PDR (Physicians Desk Reference) and the MSDS (Material Safety Data Sheet) are good ones for that. Between the two of them you pretty much have all you need to know to figure out what’s safe and what isn’t.
PDR on the internet:
https://www.pdr.net/drug-summary/Sklice-ivermectin-2415
@AnnieM, yeah, I wasn’t planning to eat them, and I don’t yet weigh 2,600 lbs :-) I was simply identifying how a cheap A$$ drug for my dog cost — 6 monthly doses = $60 bucks– for such a small dose. Please keep in mind, I grabbed a pic from the net and Zeus (my dog) is 90+ Lbs, not 26, so personal accuracy is not in the house.
I hear Pyrantel will make you grow a tail anyhow. My wife would not approve >
drugs.com – more drug info
https://www.drugs.com/
News items I see never say exactly what a person took, other than to say Ivermectin.
My purchases have always been paste in a tube for (up to) a 1,250 horse. Up until now widely avaiable for about $5.
If someone is taking a gob of this stuff without careful measurement and testing they are likely of the sort that think feeding chocolate to hens causes them to lay brown eggs.
They might understand “Don’t do that.” Said in a loud voice. But maybe not.
@Ossqss – Your dog?!?
I thought that was your office assistant and security detail, but what do I know?
I did think it was a little suspicious that all it took was a couple of hot dogs to get past your security.
😜
@H.R.:
Do note that you can NOT get past such security with roast chicken… mine just follow me around whining and howling…
@HR&EM, he does have a vulnerability for roast chicken hot dogs. I must admit, so do I :-)
“Medical Associations Citing Merck et al. Declare War on Ivermectin Use Targeting COVID-19 in America
https://trialsitenews.com/medical-associations-citing-merck-et-al-declare-war-on-ivermectin-use-targeting-covid-19-in-america/
FYI, the erythromycin group also was discovered by culturing (different) Streptomyces species back in the 1950s. If I am not mistaken, these are another class of soil microbe derived macrolides.
So sheep vomit lead to a treatment for Gail’s condition. Many therapies come from strange events, but that one probably ranks near the top!!! I am looking into it.
(ok, i know it was the meds she was giving them, but my way sounds more exotic).
I came across a helpful website compiling sources and outcomes of Ivermectin studies. There’s still lots to digest, but I thought y’all would find it interesting. Bottom line, prophylaxis Ivermectin shows an 86% improvement in outcomes vs not doing anything until you have the China virus.
https://ivmmeta.com/